Low Thyroid Condition Predisposes to Death From Infectious Disease
The greatest American Endocrinologist, Broda Barnes MD, was an astute clinician who observed the low thyroid condition is associated with reduced immunity to infectious diseases. These teachings of Dr Barnes (1906-1988) can be found in his book, Hypothyroidm, the Unsuspected Illness. Unfortunately, the mainstream endocrinologists of his time rejected Dr. Barnes teachings, including this one about hypothyroidism associated with reduced immunity.
A new study using hypothyroid mice shows that Dr Barnes was right all along.
Mouse and Infectious Disease
The ability to battle and withstand an infectious insult was studied in hypothyroid mice in 2014 by Dr Cristiana Perrotta from Milan Italy entitled,”The thyroid hormone triiodothyronine controls macrophage maturation and functions: protective role during inflammation.” (1)
In this hypothyroid mouse model,” T3 (thyroid hormone tri-iodothyronine) significantly protected mice against endotoxemia induced by lipopolysaccharide i.p. injection.”(1)
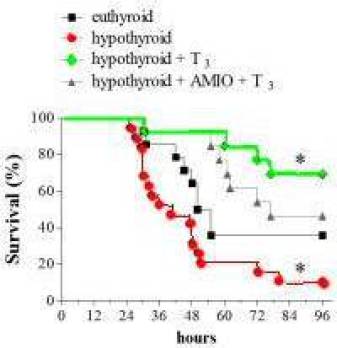
Dr Perrotta injected hypothyroid mice with gram negative bacteria lipopolysaccharide (LPS) to induce endotoxemia. The hypothyroid mice had 90% mortality after 96 hours. (RED LINE left chart) However, when the hypothyroid mice were injected with T3 (thyroid hormone) for five days before gram negative endotoxemia, they were now protected from death with 70 % survival (GREEN LINE). (see Fig 10A from Dr Perrotta (1). Notice the T3 treated mice (GREEN LINE) enjoyed the greatest survival and best outcome.
Thyroid Hormone: Protecting Us from Infection
I thought this was very impressive demonstration of the importance of thyroid hormone for boosting our immune system, and protecting us from infectious disease.
Thyroid Hormone and Cancer Immunity
Thyroid Hormone Cancer Protective in a Stress Induced Model of Lymphoma
If thyroid hormone enhances our immune system and protects us from infectious disease, one might predict thyroid hormone confers a similar protection from cancer, which after all is dependent on our immune surveillance.
This question was studied in a mouse lymphoma model by Dr Luciana Frick. The mice were subjected to chronic stress by restraining them in a narrow confinement tube. The restrained mice exhibited impaired T-cell mediated immunity and accelerated progression of lymphoma.(7,8) In addition, treatment of the mice with thyroid hormone (thyroxine) enhanced their immune systems, reversing the impaired T cell immunity, and reversing the lymphoma cell proliferation. Dr Frick concludes:
“These results show that thyroid hormones are regulators of tumor evolution, acting through the modulation of T-cell mediated immunity affected by chronic stress.”(7) ” These findings also indicate a potential therapeutic action of thyroxin in the adjuvant treatment of stress-related disorders such as immunosuppression and cancer.”
Dr Luciana Frick is in agreement with the teachings of Dr Broda Barnes, stating:
“Results presented herein are consistent with prior evidence that demonstrates that experimental hypothyroidism leads to a general depression of the immune system“(7)
Conclusion: Not only is thyroid hormone important for immune function and protection from infection disease, it is also important for immune system cancer surveillance, protecting us from cancer development.
Jeffrey Dach MD
7450 Griffin Road Suite 180
Davie Florida 33314
954-792-4663
Articles with related Interest
Ten Reasons Why You Have A Low thyroid condition
Links and references
1) Perrotta, Cristiana, et al. “The thyroid hormone triiodothyronine controls macrophage maturation and functions: protective role during inflammation.” The American journal of pathology 184.1 (2014): 230-247.
Of interest, T3 significantly protected mice against endotoxemia induced by lipopolysaccharide i.p. injection; in these damaged animals, decreased T3 levels increased the recruited (potentially damaging) cells,
The endocrine system participates in regulating macrophage maturation, although little is known about the modulating role of the thyroid hormones. In vitro results demonstrate a negative role of one such hormone, triiodothyronine (T3), in triggering the differentiation of bone marrow-derived monocytes into unpolarized macrophages. T3-induced macrophages displayed a classically activated (M1) signature. A T3-induced M1-priming effect was also observed on polarized macrophages because T3 reverses alternatively activated (M2) activation, whereas it enhances that of M1 cells. In vivo, circulating T3 increased the content of the resident macrophages in the peritoneal cavity, whereas it reduced the content of the recruited monocyte-derived cells. Of interest, T3 significantly protected mice against endotoxemia induced by lipopolysaccharide i.p. injection; in these damaged animals, decreased T3 levels increased the recruited (potentially damaging) cells, whereas restoring T3 levels decreased recruited and increased resident (potentially beneficial) cells. These data suggest that the anti-inflammatory effect of T3 is coupled to the modulation of peritoneal macrophage content, in a context not fully explained by the M1/M2 framework. Thyroid hormone receptor expression analysis and the use of different thyroid hormone receptor antagonists suggest thyroid hormone receptor β1 as the major player mediating T3 effects on macrophages. The novel homeostatic link between thyroid hormones and the pathophysiological role of macrophages opens new perspectives on the interactions between the endocrine and immune systems.
in Vivo Inflammation Experiments
As previously reported,45 and 57 acute illness was induced by a single i.p. injection of 10 to 15 μg/g body weight of LPS (endotoxin, Escherichia coli serotype O55:B5). Survival was monitored for 96 hours. In another set of experiments, mice received a single injection i.p. of LPS for 16 hours before the isolation and flow cytometry analysis of resident peritoneal cavity cells.
Thyroid Hormone TH Levels Affect Animal Mortality and Peritoneal Macrophage Subsets during Endotoxemia
LPM and SPM differ markedly in their in vivo responses to inflammatory stimuli, 56 and M1 cells (CD14+) have a classic proinflammatory profile; we evaluated whether alterations of TH levels affected the animal survival rate during systemic inflammation. To this end, we injected euthyroid and hypothyroid mice i.p. with LPS to induce endotoxemia, and we recorded the mortality of the animals over 96 hours. As shown in the Kaplan-Meier curve of Figure 10A, hypothyroid mice exhibited significantly increased mortality during the course of the experiment (10% survival of hypothyroid mice versus 35% survival of euthyroid mice). When injected i.p. with 0.2 μg/g body weight per day T3 for 5 days before the onset of endotoxemia, hypothyroid animals were significantly protected from death (70% survival).
Accordingly, we show that circulating T3 significantly protected mice against inflammation induced by LPS i.p. injection, a model of endotoxemia resembling sepsis.
relevant data on TH (thyroid hormone) levels as indicators of sepsis severity and predictors of mortality come from human studies. For instance, in patients with septic shock, tissue responses are orientated to decrease production and increase degradation (muscle) or decrease uptake (adipose tissue) of T3, as well as to decrease thyroid hormone actions.89 In addition, T3 levels were significantly lower in patients with sepsis compared with patients with an inflammatory response without underlying infection,90 and the levels of THs have been found to be a parameter for evaluating meningococcal septic shock severity. 91 and 92 A systematic review regarding the association between thyroid hormone abnormalities and the outcome of patients with sepsis or septic shock suggests the existence of an association between lower T3 or T4 and worse outcome,
2) free pdf
De Vito, Paolo, et al. “Thyroid hormones as modulators of immune activities at the cellular level.” Thyroid 21.8 (2011): 879-890.
Increasing evidence suggests that thyroid hormones, L-thyroxine (T(4)) and 3,3′,5-triiodo-L-thyronine (T(3)), are modulators of the immune response. In monocytes, macrophages, leukocytes, natural killer cells, and lymphocytes, a wide range of immune functions such as chemotaxis, phagocytosis, generation of reactive oxygen species (ROS), and cytokine synthesis and release are altered under hypo- and hyperthyroid conditions.
SUMMARY: Hyperthyroidism decreases the proinflammatory activities of monocytes and macrophages, whereas enhancement of phagocytosis and increased levels of ROS may occur during hypothyroidism. The expression of proinflammatory molecules such as macrophage inflammatory protein-1α and interleukin-1β increases in hypothyroidism. However, in Kupffer cells, proinflammatory activities such as the respiratory burst, nitric oxide synthase activity, and tumor necrosis factor-α expression may result from increased T(3) levels. Thyroid hormones also affect natural killer cell activity and cell-mediated immune responses.
Anti-Tumor Effects of T3-Receptor Complex
3) Alamino, V. A., et al. “The thyroid hormone triiodothyronine reinvigorates dendritic cells and potentiates anti-tumor immunity.” OncoImmunology 5.1 (2016): e1064579.
The immune and endocrine systems are interconnected via bidirectional networks.
normal TRβ could function as a tumor suppressor, avoiding cancer development, progression, and metastasis.3
In conclusion, our findings suggest a novel mechanism by which the T3-TRβ complex influences antitumor responses by bolstering DC-mediated T-cell activation during tumor growth.
higher concentrations of T3 and T4, within normal physiological ranges, enhance innate and adaptive immunity through maintenance of specific cell populations and greater responsiveness to immune stimuli.
4) Hodkinson, Clare F., et al. “Preliminary evidence of immune function modulation by thyroid hormones in healthy men and women aged 55–70 years.” Journal of Endocrinology 202.1 (2009): 55-63.
A reciprocal relationship between the endocrine and immune system has been demonstrated under pathophysiological conditions. However, few studies have assessed the relationship between thyroid hormones and immune function in apparently healthy individuals. Therefore, to clarify our understanding of normal physiological endocrine–immune interactions this study aimed to examine the interrelationships between thyroid hormones and immunity in healthy individuals. Total triiodothyronine (T3), total thyroxine (T4) and markers of immune status were assessed in 93 free-living and apparently healthy individuals aged 55–70 years. T3 and T4 concentrations were determined by commercially available kits. Immune status was assessed using flow cytometry and biochemical markers. Statistical analysis was performed by partial correlation, controlling for age. Thyroid hormone concentration was positively associated with markers of inflammation (P≤0.05), natural killer-like T cells (P≤0.001), expression of interleukin-6 (IL6) by activated monocytes (P≤0.05); percentage expression of memory T-lymphocytes (P≤0.01), memory T-helper lymphocytes (P≤0.05) and memory T-cytotoxic lymphocytes (P≤0.05), and higher IL2 receptor density on CD3+T-lymphocytes (P≤0.05). Thyroid hormone concentration was inversely associated with early lymphocyte apoptosis (P≤0.05) and the ratio of naïve- to memory T-cytotoxic lymphocytes (P≤0.05). The current study provides preliminary evidence of a role for T3 and T4, within normal physiological ranges, in the maintenance of lymphocyte subpopulations, and in mediating the inflammatory response. In conclusion, these findings highlight the potential implications of altered thyroid function in older individuals and the importance of future research examining thyroid–immune interactions.
To our knowledge this is the first study to examine associations between thyroid hormone concentration and markers of immunity in healthy men and women, using a comprehensive panel of immune markers. Thyroid hormone concentration and markers of immunity were within normal physiological ranges. Overall, the current study provides preliminary evidence to suggest that higher concentrations of T3 and T4, within normal physiological ranges, enhance innate and adaptive immunity through maintenance of specific cell populations and greater responsiveness to immune stimuli.
These preliminary findings indicate the potential benefit of maintaining optimal thyroid hormone concentrations for the maintenance of immune function with advancing age.
5)
free pdf
G Ahmed, R., et al. “Nongenomic actions of thyroid hormones: from basic research to clinical applications. An update.” Immunology, Endocrine & Metabolic Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Immunology, Endocrine and Metabolic Agents) 13.1 (2013): 46-59.
Thyroid hormones may have an important role also in neuroprotection.
IMMUNE SYSTEM CELLS
The last few years have brought increasing evidence con-firming the importance of thyroid hormones in immune sys-tem function and physiology [8, 9, 76, 77]. In general, hyper-
thyroidism enhances the immune response in terms of anti-body production, cell migration, lymphocyte proliferation and reactive oxygen species (ROS) production, whereas it is associated with decreased proinflammatory marker release, antioxidant enzyme production and immune functions [9]. Hypothyroidism often gives rise to the opposite effects for some of these parameters: it decreases the immune response, antibody production, cell migration and lymphocyte prolif- eration [77, 78].
anti-viral state
Thyroid hormones are able to potentiate the antiviral state induced by IFN-gamma, a pro-inflammatory and pro-apoptotic cytokine that regulates host defense, on HLA-DR
expression in CV-1 fibroblasts and HeLa cells, these cells are devoid of the classical nuclear receptors and therefore any effects observed must be due to hormone binding at the
plasma membrane or in the cytosol [11].
6) Kim, Won Gu, and Sheue-yann Cheng. “Thyroid hormone receptors and cancer.” Biochimica et Biophysica Acta (BBA)-General Subjects 1830.7 (2013): 3928-3936.
Loss of normal functions of TR by deletion or by mutations could contribute to cancer development, progression and metastasis.
!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
7) free pdf
Frick, Luciana Romina, et al. “Involvement of thyroid hormones in the alterations of T-cell immunity and tumor progression induced by chronic stress.” Biological psychiatry 65.11 (2009): 935-942.
Stress alters the neuroendocrine system, immunity, and cancer. Although the classic stress hormones are glucocorticoids and catecholamines, thyroid hormones have also been related to stress. We recently reported that chronic restraint stress impairs T-cell mediated immunity and enhances tumor growth in mice.
METHODS:To study the participation of these hormones on the stress-induced alterations of the immune function and lymphoma growth, mice were subjected to acute or chronic stress, with or without thyroxin supplementation. Hormone levels, immune status, and cancer progression were evaluated.
RESULTS:Differential endocrine alterations were observed in response to acute and chronic stress. Although corticosterone and noradrenaline levels were increased by acute stress, they were restored after prolonged exposure to the stressor. Instead, thyroid hormone levels were only reduced in chronically stressed animals in comparison with control subjects. Correlating, chronic but not acute stress impaired T-cell reactivity. Thyroxin replacement treatment of chronic restraint stress-exposed mice, which restored the euthyroid status, reversed the observed reduction of T-cell lymphoproliferative responses. Moreover, therapeutic thyroid replacement also reversed the alterations of lymphoma growth induced by chronic stress in syngeneic mice bearing tumors as well as Interleukin-2 production and specific cytotoxic response against tumor cells. Finally, we found that the isoforms theta and alpha of the protein kinase C are involved in these events.
CONCLUSIONS:These results show for the first time that thyroid hormones are important neuroendocrine regulators of tumor evolution, most probably acting through the modulation of T-cell mediated immunity affected by chronic stress.
(i.e., hypo-thyroidism impairs whereas hyperthyroidism enhances T and B lymphocyte responses)
In a recent work, we showed that chronic restraint stress selectively impairs T-cell mediated immunity, which is accompa-nied by an accelerated progression of a syngeneic lymphoma
model (26).
To our knowledge, this is the first study to demonstrate a relationship between stress, thyroid axis, immunity, and cancer
Results presented herein are consistent with prior evi-dence that demonstrates that experimental hypothyroidism leads to a general depression of the immune system (12,13).
Strengthening our hypothesis of a relation between thyroid hormones and lymphoma progression, the reduction of T3 and T4 levels was also observed in chronically stressed mice bearing solid tumors. Thyroxin replacement in these animals prevented the exacerbation of tumor growth and the inhibition of cytotoxic response against lymphoma cells as well as of cytokine expres-sion.
Taken together, the present results show for the first time that thyroid hormones are implicated in the impairment of T-cell– mediated immunity and the enhancement of tumor progression induced by chronic restraint stress in a murine model of lym- phoma . These findings also indicate a potential therapeutic action of thyroxin in the adjuvant treatment of stress-related disorders such as immunosuppression and cancer
————————
stress promotes lymphoma
8) Frick, L. R., et al. “Chronic restraint stress impairs T-cell immunity and promotes tumor progression in mice.” Stress 12.2 (2009): 134-143.
Chronic restraint stress impairs T-cell immunity and promotes tumor progression in mice.
Frick LR1, Arcos ML, Rapanelli M, Zappia MP, Brocco M, Mongini C, Genaro AM, Cremaschi GA.
Long-term exposure to stressful situations can affect the immune system. The T-cell response is an important component of anti-tumoral immunity. Hence, impairment of the immune function induced by a chronic stressor has been postulated to alter the immunosurveillance of tumors, thus leading to a worse neoplastic prognosis. Here, we show that chronic restraint stress affects T-cell mediated immunity in mice. This was evidenced by a decrease of mitogen-induced T-cell proliferation, a reduction in CD4(+)T lymphocyte number and a decrease of tumor necrosis factor-alpha (TNF-alpha) and Interferon-gamma (IFN-gamma) production in stressed mice. Additionally, mice subjected to chronic restraint stress displayed an enhancement of tumor growth in a syngeneic lymphoma model, i.e. an increase of tumor proliferation and a reduction of animal survival. Finally, stressed mice had a reduced specific cytotoxic response against these tumor cells. These results suggest that chronic exposure to stress promotes cancer establishment and subsequent progression, probably by depressing T-cell mediated immunity. The T-cell immunity impairment as well as the tumor progression enhancement emphasize the importance of the therapeutic management of stress to improve the prognosis of cancer patients.
9) Klecha, Alicia J., et al. “Integrative study of hypothalamus–pituitary–thyroid–immune system interaction: thyroid hormone-mediated modulation of lymphocyte activity through the protein kinase C signaling pathway.” Journal of Endocrinology 189.1 (2006): 45-55.
Thyroid hormones play critical roles in differentiation, growth and metabolism, but their participation in immune system regulation has not been completely elucidated. Modulation of in vivo thyroid status was used to carry out an integrative analysis of the role of the hypothalamus-pituitary-thyroid (HPT) axis in T and B lymphocyte activity. The participation of the protein kinase C (PKC) signaling pathway and the release of some cytokines upon antigenic stimulation were analyzed. Lymphocytes from hyperthyroid mice displayed higher T-and B-cell mitogen-induced proliferation, and those from hypothyroid mice displayed lower T- and B-cell mitogen-induced proliferation, compared with euthyroid animals. Reversion of hypothyroid state by triiodothyronine (T3) administration recovered the proliferative responses. No differences were found in lymphoid subset balance. Both total PKC content and mitogen-induced PKC translocation were higher in T and B cells from hyperthyroid mice, and lower in cells from hypothyroid mice, compared with controls. Levels of thyroid-stimulating (TSH) and TSH-releasing (TRH) hormones were not directly related to lymphocyte proliferative responses. After immunization with sheep red blood cells (SRBCs) and re-stimulation, in vitro spleen cells from hyper- or hypothyroid mice showed, respectively, increased or decreased production of interleukin (IL)-2 and interferon (IFN)-gamma cytokines. Additionally, an increase in IL-6 and IFN-gamma levels was found in hyperthyroid cells after in vivo injection and in vitro re-stimulation with lipopolysaccharide (LPS). Our results show for the first time a thyroid hormone-mediated regulation of PKC content and of cytokine production in lymphocytes; this regulation could be involved in the altered responsiveness to mitogen-induced proliferation of T and B cells. The results also confirm the important role that these hormones play in regulating lymphocyte reactivity.
10) Schoenfeld, Philip S., et al. “Suppression of cell-mediated immunity in hypothyroidism.” Southern medical journal 88.3 (1995): 347-349.
A 71-year-old man had severe hypothyroidism, chronic autoimmune thyroiditis, and bacteremia due to Edwardsiella tarda. Review of the literature identified the hypothesis that E tarda infections may occur more frequently in immunocompromised patients. Previous animal studies have shown decreases in lymphocyte function during hypothyroidism, with return of normal lymphocyte function during euthyroid states. Therefore, lymphocyte transformation studies were obtained, demonstrating severe decreases in our patient’s lymphocyte function. Except for chronic autoimmune thyroiditis, other immune system abnormalities were excluded. Serial lymphocyte transformation studies showed gradual improvement in lymphocyte function during gradual return to euthyroid state. Possible pathophysiologic mechanisms for these findings will be reviewed.
Link to this article:http://wp.me/p3gFbV-3SW
Jeffrey Dach MD
7450 Griffin Road, Suite 180
Davie, Fl 33314
954-972-4663
www.jeffreydachmd.com
www.drdach.com
www.naturalmedicine101.com
www.bioidenticalhormones101.com
www.truemedmd.com
Web Site and Discussion Board Links:
jdach1.typepad.com/blog/
disc.yourwebapps.com/Indices/244124.html
disc.yourwebapps.com/Indices/244066.html
disc.yourwebapps.com/Indices/244067.html
disc.yourwebapps.com/Indices/244161.html
disc.yourwebapps.com/Indices/244163.html
Disclaimer click here: www.drdach.com/wst_page20.html
The reader is advised to discuss the comments on these pages with his/her personal physicians and to only act upon the advice of his/her personal physician. Also note that concerning an answer which appears as an electronically posted question, I am NOT creating a physician — patient relationship. Although identities will remain confidential as much as possible, as I can not control the media, I can not take responsibility for any breaches of confidentiality that may occur.
Copyright (c) 2016 Jeffrey Dach MD All Rights Reserved. This article may be reproduced on the internet without permission, provided there is a link to this page and proper credit is given.
FAIR USE NOTICE: This site contains copyrighted material the use of which has not always been specifically authorized by the copyright owner. We are making such material available in our efforts to advance understanding of issues of significance. We believe this constitutes a ‘fair use’ of any such copyrighted material as provided for in section 107 of the US Copyright Law. In accordance with Title 17 U.S.C. Section 107, the material on this site is distributed without profit to those who have expressed a prior interest in receiving the included information for research and educational purposes.
Serving Areas of: Hollywood, Aventura, Miami, Fort Lauderdale, Pembroke Pines, Miramar, Davie, Coral Springs, Cooper City, Sunshine Ranches, Hallandale, Surfside, Miami Beach, Sunny Isles, Normandy Isles, Coral Gables, Hialeah, Golden Beach ,Kendall,sunrise, coral springs, parkland,pompano, boca raton, palm beach, weston, dania beach, tamarac, oakland park, boynton beach, delray,lake worth,wellington,plantation.
The post Hypothyroid Risks for Infection appeared first on Jeffrey Dach MD .









Leave a Comment
You must be logged in to post a comment.