Clostridia Difficile Enterocolitis Causes Prevention and Treatment
by Jeffrey Dach MD
If you wake up one morning in a hospital bed, you might want to know your risk of C Diff (Clostridia Enterocolitis) is increased if the previous occupant was given antibiotics, according to a study in JAMA.(1) Left image Clostridia Difficile Electron Microscope Image courtesy of the CDC.
IV Antibiotics Anyone?
Of course, the use of IV antibiotics is now recognized as the single most significant risk factor for C Diff (clostridia difficile enterocolitis) in the hospital setting.(5) The antibiotics obliterate the friendly bacteria in the gut, setting the backdrop for overgrowth of pathogenic Clostridia Difficile. More virulent strains of the organism have been making headway with exponential increase in cases on the surgical wards.
C Diff Cases Increasing on the Surgical Ward
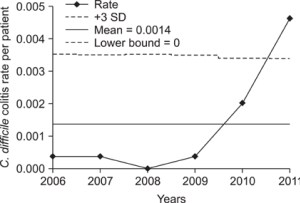 Left chart shows exponential increase in C Diff on the surgical wards. Antibiotics anyone? Courtesy of Kim et al 2012.
Left chart shows exponential increase in C Diff on the surgical wards. Antibiotics anyone? Courtesy of Kim et al 2012.
The Chemotherapy Patient and C. Diff
The chemotherapy patient may be at increased risk for C Diff enterocolitis for a number of reasons. Leukemia and lymphoma patients are commonly treated with antibiotics after chemotherapy. Chemotherapy not only acts an antibiotic disrupting the gut flora. Chemotherapy may also disrupt the mucus barrier function of the gut as well, all predisposing to leaky gut and gram negative septicemia (also known as endotoxemia)
Intensive chemotherapy protocols cause bone marrow suppression resulting in very low white cell counts and immuno-suppression which places the patient at risk for infection. Indeed febrile neutropenia and hypotension is common after high dose chemotherapy, indicating gram negative septicemia, a life threatening medical emergency requiring hospitalization and treatment with massive IV antibiotics and pressors to maintain the blood pressure. If the patient is among the lucky few to survives such a medical mishap, the massive use of IV antibiotics increase the risk for subsequent C Diff enterocolitis.
C Diff is a more severe problem in the hematopoetic stem cell transplant ward where recurrent C Diff can increase the mortality of the procedure due to infection and graft vs host disease. (11), The use of preventive probiotics may prove useful in the stem cell transplant setting for improving overall survival, and reducing non-relapse mortality. See my previous article on “Probiotics Reduce Mortality for the Stem Cell Transplant Patient.”
Significant Mortality Rate Goes Up with Recurrence
The common hospital acquired infection, Clostridia Difficile has a mortality rate of about 8-9%, with half a million infections and 29,000 deaths in 2011.(2) The incidence has been increasing rapidly. Pathogenic C. Diff produces two toxins which cause watery diarrhea, and electrolyte disturbance. In severe cases there may be pseudo-membranous colitis and toxic megacolon requiring heroic measures such as total colectomy.
The C Diff infection usually responds to antibiotics such as Vancomycin and metronidazole (Flagyl). However, antibiotics produce further disturbance in the gut microbiota, and fail to eradicate the spore form of C Diff, explaining the 30%-50% recurrence rate, an outcome associated with increased mortality. (3-4)
In-house hospital prevention of C Diff relies on standard infectious disease techniques such as microbial isolation, and hand washing before and after donning masks, gloves and gowns etc. Clearly, there is a need for improved prevention and treatment of C Diff enteritis in the hospital setting.
Preventing C Diff with Probiotics
Donna L. Parker RN, director of nursing for a nursing residence experienced a high rate of C Diff among her patients treated with antibiotics. After instituting a policy of giving a probiotic tablet along with the antibiotics, she reports no cases over two years. She writes about her experience using probiotics to prevent C Diff in her October 2016 article, “Preventing C. diff infections with probiotics”. (10)
Others have studied the use of probiotics such as Lactobacillus GG and Sacchromyces Boulardi, and have found them effective in preventing C Diff.(7-13)
“L. rhamnosus GG has been shown to increase gut mucin production [Mack et al. 1999], which improves the barrier defences of the epithelium, and increases colonic water absorption [Madsen et al. 2001], which directly reduces diarrhoea.” (13) (Mary Hickson)
Recommended Probiotics
Renew Life 200 Billion Packets
Lactobacillus Rhamnosus GG Culturelle 10B
Miyarisan Clostridium Butyricum Nontoxigenic strain
Kefir as Probiotic
Kefir, containing multiple lactobacillus strains of probiotic grown in a milk substrate is a popular item at the grocery store, and available as a home starter kit. A number of studies show health benefits from Kefir consumption.(14-15) Surprisingly, kefir was found to have significant anti-cancer activity in a mouse cancer xenograft model. (15)
Preventing Recurrence with Berberine
The botanical, berberine, also known as the Orgegon Grape, has been used for thousands of years effective for watery diarhea. Studies show that berberine capsules taken together with vancomycin prevent recurrence of Clostridia Difficile. (6) See my previous article on Berberine as an antidote to a modern epidemic .
Preventing Recurrent C Diff With Fecal Transplant
As mentioned in my previous article on Fecal Transplantation, this procedure is actually FDA approved for the treatment of recurrent C Diff. in over 100 US hospitals. (16) After all, one might regard fecal transplantation as simply another way to administer a massive dose of probiotics.
Preventing Recurrent C Diff with Niclosamide
The anti-parasitic drug Niclosamide is FDA approved in the US for treatment of tapeworm, and has been repurposed for treatment of C Diff. (17-18) The added advantage of Niclosamide is this drug will eradicate the spore form of C Diff resistant to conventional antibiotics. In addition, unlike conventional antibiotics which disturb the normal flora, Niclosamide leaves the normal flora relatively unharmed. An added bonus for cancer patients, Niclosamide has potent anti-cancer activity at conventional dosing schedules. (19)
Conclusion:
Although mainstream medicine may be slow to adopt new ideas, improved strategies to prevent and treat C Diff are already available. The only thing left to do is use them.
Jeffrey Dach MD
7450 Griffin Road Suite 190
Davie, Fl 33314
954 792 4663
Articles with Related Interest:
Probiotics for the Stem Cell Transplant Patient
Berberine Antidote to an Epidemic
Fecal Transplantation, The Sweet Smell of Success
1) JAMA Less Is More October 10, 2016
Receipt of Antibiotics in Hospitalized Patients and Risk for Clostridium difficile Infection in Subsequent Patients Who Occupy the Same Bed
Daniel E. Freedberg, MD, MS1; Hojjat Salmasian, MD, PhD2; Bevin Cohen, MPH3; et al Julian A. Abrams, MD, MS1; Elaine L. Larson, RN, PhD3
1Division of Digestive and Liver Diseases, Columbia University Medical Center, New York, New York
2Department of Biomedical Informatics, New York-Presbyterian Hospital, New York, New York
3Department of Epidemiology, Mailman School of Public Health, School of Nursing, Columbia University, New York, New York
Objective To assess whether receipt of antibiotics by prior hospital bed occupants is associated with increased risk for CDI in subsequent patients who occupy the same bed.
Design, Setting, and Participants This is a retrospective cohort study of adult patients hospitalized in any 1 of 4 facilities between 2010 and 2015. Patients were excluded if they had recent CDI, developed CDI within 48 hours of admission, had inadequate follow-up time, or if their prior bed occupant was in the bed for less than 24 hours.
Main Outcomes and Measures The primary exposure was receipt of non-CDI antibiotics by the prior bed occupant and the primary outcome was incident CDI in the subsequent patient to occupy the same bed. Incident CDI was defined as a positive result from a stool polymerase chain reaction for the C difficile toxin B gene followed by treatment for CDI. Demographics, comorbidities, laboratory data, and medication exposures are reported.
Results Among 100 615 pairs of patients who sequentially occupied a given hospital bed, there were 576 pairs (0.57%) in which subsequent patients developed CDI. Receipt of antibiotics in prior patients was significantly associated with incident CDI in subsequent patients (log-rank P < .01). This relationship remained unchanged after adjusting for factors known to influence risk for CDI including receipt of antibiotics by the subsequent patient (adjusted hazard ratio [aHR], 1.22; 95% CI, 1.02-1.45) and also after excluding 1497 patient pairs among whom the prior patients developed CDI (aHR, 1.20; 95% CI, 1.01-1.43). Aside from antibiotics, no other factors related to the prior bed occupants were associated with increased risk for CDI in subsequent patients.
Conclusions and Relevance Receipt of antibiotics by prior bed occupants was associated with increased risk for CDI in subsequent patients. Antibiotics can directly affect risk for CDI in patients who do not themselves receive antibiotics.
2) Burden of Clostridium difficile Infection in the United States Fernanda C. Lessa, M.D., M.P.H., et al. N Engl J Med 2015; 372:825-834 February 26, 2015
C. difficile was responsible for almost half a million infections and was associated with approximately 29,000 deaths in 2011
3) Olsen, Margaret A., et al. “Recurrent Clostridium difficile infection is associated with increased mortality.” Clinical Microbiology and Infection 21.2 (2015): 164-170. Clostridium difficile infections (CDI) are associated with decreased survival, and up to 30% of CDI patients may experience a recurrence. Data on the impact of recurrent CDI on mortality are scarce. The purpose of this study was to determine whether recurrent CDI was independently associated with decreased 6-month survival compared with patients with CDI who did not develop a recurrence. We performed a retrospective cohort study at an academic, urban, tertiary care hospital. Data were collected from the electronic medical record and chart review. CDI patients were followed for 180 days from the end of their index hospital discharge or end of index CDI antibiotic treatment, whichever was later, to determine mortality. Kaplan–Meier analysis was used to compare patient mortality by recurrent CDI status. Cox proportional hazards models were used to determine independent risk factors for death within 180 days. In all, 3958 patients aged ≥ 18 years who developed an initial CDI episode from 2003 to 2009, including 421 patients with recurrent CDI, were included in the study. Thirty-six per cent of persons with recurrent CDI died within 180 days, compared with 26% of persons without CDI recurrence (log-rank p <0.001). Recurrent CDI was associated with significantly higher hazards of death within 180 days, adjusting for demographics, comorbidities and medications received during the index CDI hospitalization (hazard ratio 1.33; 95% CI 1.12–1.58). Recurrent CDI is associated with significantly increased risk of death within 6 months after completion of their initial CDI treatment compared with CDI patients who do not develop a recurrence.
4) Deshpande, Abhishek, et al. “Risk factors for recurrent Clostridium difficile infection: a systematic review and meta-analysis.” infection control & hospital epidemiology 36.04 (2015): 452-460.
5) Pépin, Jacques, et al. “Emergence of fluoroquinolones as the predominant risk factor for Clostridium difficile–associated diarrhea: a cohort study during an epidemic in Quebec.” Clinical Infectious Diseases 41.9 (2005): 1254-1260.
Since 2002, an epidemic of Clostridium difficile–associated-diarrhea (CDAD) associated with a high case-fatality rate has involved >30 hospitals in the province of Quebec, Canada. In 2003, a total of 55% of patients with CDAD at our hospital had received fluoroquinolones in the preceding 2 months. It has been suggested that massive use of proton pump inhibitors might have facilitated this epidemic.
Methods. To delineate the risk of CDAD associated with specific classes of antibiotics and whether this is modulated by concomitant use of proton pump inhibitors and other drugs altering gastric acidity or gastrointestinal motility, we conducted a retrospective cohort study of patients hospitalized in a teaching hospital in Sherbrooke, Canada, during the period of January 2003 through June 2004. We obtained data on 7421 episodes of care corresponding to 5619 individuals. Patients were observed until they either developed CDAD or died or for 60 days after discharge from the hospital. Adjusted hazard ratios (AHRs) were calculated using Cox regression.
Results. CDAD occurred in 293 patients. Fluoroquinolones were the antibiotics most strongly associated with CDAD (AHR, 3.44; 95% confidence interval [CI], 2.65–4.47). Almost one-fourth of all inpatients received quinolones, for which the population-attributable fraction of CDAD was 35.9%. All 3 generations of cephalosporins, macrolides, clindamycin, and intravenous β-lactam/β-lactamase inhibitors were intermediate-risk antibiotics, with similar AHRs (1.56–1.89). Proton pump inhibitors (AHR, 1.00, 95% CI, 0.79–1.28) were not associated with CDAD.
Conclusions. Administration of fluoroquinolones emerged as the most important risk factor for CDAD in Quebec during an epidemic caused by a hypervirulent strain of C. difficile.
Berberine
6) Antimicrob Agents Chemother. 2015 Jul;59(7):3726-35.
Berberine blocks the relapse of Clostridium difficile infection in C57BL/6 mice after standard vancomycin treatment.
Lv Z1, Peng G2, Liu W3, Xu H4, Su J5.
Vancomycin is a preferred antibiotic for treating Clostridium difficile infection (CDI) and has been associated with a rate of recurrence of CDI of as high as 20% in treated patients. Recent studies have suggested that berberine, an alternative medical therapy for gastroenteritis and diarrhea, exhibits several beneficial effects, including induction of anti-inflammatory responses and restoration of the intestinal barrier function. This study investigated the therapeutic effects of berberine on preventing CDI relapse and restoring the gut microbiota in a mouse model. Berberine was administered through gavage to C57BL/6 mice with established CDI-induced intestinal injury and colitis. The disease activity index (DAI), mean relative weight, histopathology scores, and levels of toxins A and B in fecal samples were measured. An Illumina sequencing-based analysis of 16S rRNA genes was used to determine the overall structural change in the microbiota in the mouse ileocecum. Berberine administration significantly promoted the restoration of the intestinal microbiota by inhibiting the expansion of members of the family Enterobacteriaceae and counteracting the side effects of vancomycin treatment. Therapy consisting of vancomycin and berberine combined prevented weight loss, improved the DAI and the histopathology scores, and effectively decreased the mortality rate. Berberine prevented CDIs from relapsing and significantly improved survival in the mouse model of CDI. Our data indicate that a combination of berberine and vancomycin is more effective than vancomycin alone for treating CDI. One of the possible mechanisms by which berberine prevents a CDI relapse is through modulation of the gut microbiota. Although this conclusion was generated in the case of the mouse model, use of the combination of vancomycin and berberine and represent a novel therapeutic approach targeting CDI.
7) Nat Rev Gastroenterol Hepatol. 2016 Mar;13(3):150-60. doi: 10.1038/nrgastro.2015.220. Epub 2016 Feb 10.
Breakthroughs in the treatment and prevention of Clostridium difficile infection. Kociolek LK1, Gerding DN2.
This Review summarizes the latest advances in the treatment and prevention of Clostridium difficile infection (CDI), which is now the most common health-care-associated infection in the USA. As traditional, standard CDI antibiotic therapies (metronidazole and vancomycin) are limited by their broad spectrum and further perturbation of the intestinal microbiota, which result in unacceptably high recurrence rates, novel therapeutic strategies for CDI are needed. Emerging CDI therapies are focused on limiting further perturbation of the intestinal microbiota and/or restoring the microbiota to its pre-morbid state, reducing colonization of the intestinal tract by toxigenic strains of C. difficile and bolstering the host immune response against C. difficile toxins. Fidaxomicin is associated with reduced CDI recurrences, and other emerging narrow-spectrum CDI antibiotic therapies might eventually demonstrate a similar benefit. Prevention of intestinal colonization of toxigenic strains of C. difficile can be achieved through restoration of the intestinal microbiota with faecal microbiota transplantation, as well as by colonizing the gut with nontoxigenic C. difficile strains. Finally, emerging immunological therapies, including monoclonal antibodies and vaccines against C. difficile toxins, might protect against CDI and subsequent CDI recurrences. The available clinical data for these emerging therapies, and their relative advantages and disadvantages, are described.
8) Int J Gen Med. 2016; 9: 27–37.
Probiotics are effective at preventing Clostridium difficile-associated diarrhea: a systematic review and meta-analysis
Christine SM Lau1,2 and Ronald S Chamberlain1,2,3 Probiotic supplementation is associated with a significant reduction in the risk of developing CDAD in patients receiving antibiotics.
9) Evans, Charlesnika T., and Stuart Johnson. “Prevention of Clostridium difficile infection with probiotics.” Clinical Infectious Diseases 60.suppl 2 (2015): S122-S128.
10) Donna L. Parker, RN October 06, 2016 Preventing C. diff infections with probiotics
Chemotherapy:
11) Clostridium difficile Infection Associated with Antineoplastic Chemotherapy: A Review. Ajay Anand and Aaron E. Glatt From the Department of Medicine, Nassau County Medical Center, East Meadow; the Division of Infectious Diseases, Department of Medicine, Catholic Medical Center, Jamaica; and SUNY Health Sciences Center at Stony Brook, Stony Brook, New York
Colitis and infection due to Clostridium difficile have been reported in patients receiving antineoplastic chemotherapy for cancer without prior antibiotic treatment. Chemotherapeutic agents can alter the normal bowel flora and cause extensive intestinal inflammatory changes, potentiating both the growth of C. difficile and its production of toxin. This review includes all 23 known reported cases of C. difficile infection associated with antineoplastic chemotherapy and examines the pathogenesis, clinical features, and management of this condition. Chemotherapy-associated C. difficile colitis has been documented in association with a variety of neoplasms. Various classes of antineoplastic agents have been incriminated, methotrexate most commonly. A spectrum of illness ranging from mild to fulminant has been reported. Symptoms, management, and outcome have appeared to be no different than for antibiotic-associated cases, but the available data are limited. Chemotherapy-associated infection with C. difficile may be underreported because it is not suspected and/or because frequent concomitant use of antibiotics masks its true incidence. C. difficile infection should be kept in mind whenever a patient undergoing antineoplastic chemotherapy develops diarrhea. Prompt, appropriate diagnostic testing and early treatment may avert morbidity and death.
Saccharomyces boulardii:
12) Prevention of Clostridium difficile infection with Saccharomyces boulardii: A systematic review
Jennifer M Tung, BSc,1 Lisa R Dolovich, BSc MSc,1 and Christine H Lee, MD FRCPC1,2
Clostridium difficile is a major cause of antibiotic associated diarrhea within the hospital setting. The yeast Saccharomyces boulardii has been found to have some effect in reducing the risk of C difficile infection (CDI); however, its role in preventive therapy has yet to be firmly established.
OBJECTIVE:To review the effectiveness of S boulardii in the prevention of primary and recurrent CDI. Benefit was defined as a reduction of diarrhea associated with C difficile. Risk was defined as any adverse effects of S boulardii.
METHODS:A literature search in MEDLINE, EMBASE, CINAHL and the Cochrane Library was performed. Included studies were English language, randomized, double-blind placebo controlled trials evaluating S boulardii in CDI prevention.
RESULTS:Four studies were reviewed. Two studies investigated the prevention of recurrence in populations that were experiencing CDI at baseline. One trial showed a reduction of relapses in patients experiencing recurrent CDI (RR=0.53; P<0.05). The other demonstrated a trend toward reduction of CDI relapse in the recurrent treatment group of patients receiving high-dose vancomycin (RR=0.33; P=0.05). Two other studies examined primary prevention of CDI in populations that had been recently prescribed antibiotics. These studies lacked the power to detect statistically significant differences. Patients on treatment experienced increased risk for thirst and constipation.
CONCLUSION:S boulardii seems to be well tolerated and may be effective for secondary prevention in some specific patient populations with particular concurrent antibiotic treatment. Its role in primary prevention is poorly defined and more research is required before changes in practice are recommended.
13) Therap Adv Gastroenterol. 2011 May; 4(3): 185–197.
Probiotics in the prevention of antibiotic-associated diarrhoea and Clostridium difficile infection Mary Hickson
Diarrhoea, as a common side effect of antibiotics, increases treatment costs and length of stay in acute healthcare facilities. One potential strategy to prevent this side effect is the concurrent use of probiotic bacteria or yeast. This review discusses the evidence for the efficacy of probiotics in the prevention of antibiotic-associated diarrhoea and Clostridium difficile infection; the potential mechanisms by which probiotics may work; their safety; what future research is required; and recommendations for use in clinical practice.
L. rhamnosus GG has been shown to increase gut mucin production [Mack et al. 1999], which improves the barrier defences of the epithelium, and increases colonic water absorption [Madsen et al. 2001], which directly reduces diarrhoea.
Kefir- Probiotic Foods
14) Lett Appl Microbiol. 2002;35(2):136-40.
Dietary influence of kefir on microbial activities in the mouse bowel.
Marquina D1, Santos A, Corpas I, Muñoz J, Zazo J, Peinado JM.
In this work the microflora present in kefir, a fermented milk product, was studied together with the effect of kefir administration on different groups of indigenous bacteria of mouse bowel.
METHODS AND RESULTS: Kefir microflora was composed of lactic acid bacteria, acetic acid bacteria and yeasts. Yeast population was composed of Saccharomyces cerevisiae, S. unisporus, Candida kefir, Kluyveromyces marxianus and K. lactis. The streptococci levels in kefir treated mice increased by 10-fold and the levels of sulfite-reducing clostridia decreased by 100-fold. The number of lactic acid bacteria increased significantly.
CONCLUSIONS: The administration of kefir significantly increased the lactic acid bacteria counts in the mucosa of the bowel. Ingestion of kefir specifically lowered microbial populations of Enterobacteriaceae and clostridia.
SIGNIFICANCE AND IMPACT OF THE STUDY: This is the first long-term study about the effects of the kefir administration on the intestinal microflora of mice.
15) Antitumor Activity of Milk Kefir and Soy Milk Kefir in Tumor-Bearing Mice Je-Ruei Liu , Sheng-Yao Wang , Yuh-Yih Lin & Chin-Wen Lin
Pages 183-187 | Published online: 18 Nov 2009
Abstract: The effects of oral administration of milk and soy milk kefirs on tumor growth in tumor-bearing mice and the mucosal immunoglobulin A response in mice were studied. Oral administration of milk and soy milk kefirs to mice inoculated with sarcoma 180 tumor cells resulted in 64.8% and 70.9% inhibition of tumor growth, respectively, compared with controls. In addition, oral administration of the two kefir types induced apoptotic tumor cell lysis. Total immunoglobulin A levels for tissue extracts from the wall of the small intestine were also significantly higher for mice fed a milk kefir or a soy milk kefir regimen for 30 days. These results suggest that milk and soy milk kefirs may be considered among the more promising food components in terms of cancer prevention and enhancement of mucosal resistance to gastrointestinal infection.
Fecal Transplant
16)
Drekonja, Dimitri, et al. “Fecal microbiota transplantation for Clostridium difficile infection: a systematic review.” Annals of internal medicine 162.9 (2015): 630-638.
Niclosamide
17) Reprofiled anthelmintics abate hypervirulent stationary-phase Clostridium difficile . Major Gooyit & Kim D. Janda Scientific Reports 6, Article number: 33642 (2016)
Prolonged use of broad-spectrum antibiotics disrupts the indigenous gut microbiota, which consequently enables toxigenic Clostridium difficile species to proliferate and cause infection. The burden of C. difficile infections was exacerbated with the outbreak of hypervirulent strains that produce copious amounts of enterotoxins and spores. In recent past, membrane-active agents have generated a surge of interest due to their bactericidal property with a low propensity for resistance. In this study, we capitalized on the antimicrobial property and low oral bioavailability of salicylanilide anthelmintics (closantel, rafoxanide, niclosamide, oxyclozanide) to target the gut pathogen. By broth microdilution techniques, we determined the MIC values of the anthelmintics against 16 C. difficile isolates of defined PCR-ribotype. The anthelmintics broadly inhibited C. difficile growth in vitro via a membrane depolarization mechanism. Interestingly, the salicylanilides were bactericidal against logarithmic- and stationary-phase cultures of the BI/NAP1/027 strain 4118. The salicylanilides were poorly active against select gut commensals (Bacteroides, Bifidobacterium and Lactobacillus species), and were non-hemolytic and non-toxic to mammalian cell lines HepG2 and HEK 293T/17 within the range of their in vitro MICs and MBCs. The salicylanilide anthelmintics exhibit desirable properties for repositioning as anti-C. difficile agents.
18) Clostridium difficile Drug Pipeline: Challenges in Discovery and Development of New Agents. Angie M. Jarrad†, Tomislav Karoli†, Mark A. T. Blaskovich†, Dena Lyras‡, and Matthew A. Cooper*†
† The Institute for Molecular Bioscience, University of Queensland, St. Lucia, Queensland 4072, Australia
‡ School of Biomedical Sciences, Monash University, Clayton, Victoria 3800, Australia. J. Med. Chem., 2015, 58 (13), pp 5164–5185
In the past decade Clostridium difficile has become a bacterial pathogen of global significance. Epidemic strains have spread throughout hospitals, while community acquired infections and other sources ensure a constant inoculation of spores into hospitals. In response to the increasing medical burden, a new C. difficile antibiotic, fidaxomicin, was approved in 2011 for the treatment of C. difficile-associated diarrhea. Rudimentary fecal transplants are also being trialed as effective treatments. Despite these advances, therapies that are more effective against C. difficile spores and less damaging to the resident gastrointestinal microbiome and that reduce recurrent disease are still desperately needed. However, bringing a new treatment for C. difficile infection to market involves particular challenges. This review covers the current drug discovery pipeline, including both small molecule and biologic therapies, and highlights the challenges associated with in vitro and in vivo models of C. difficile infection for drug screening and lead optimization.
19) Cancer Lett. 2014 Jul 10; 349(1): 8–14.
Multi-targeted therapy of cancer by niclosamide: a new application for an old drug Yonghe Li,1,* Pui Kai Li,2 Michael J. Roberts,3 Rebecca Arend,4 Rajeev S. Samant,5 and Donald J. Buchsbaum6
The rapid development of new anticancer drugs that are safe and effective is a common goal shared by basic scientists, clinicians and patients. The current review discusses one such agent, namely niclosamide, which has been used in the clinic for the treatment of intestinal parasite infections. Recent studies repeatedly identified niclosamide as a potential anticancer agent by various high-throughput screening campaigns. Niclosamide not only inhibits the Wnt/β-catenin, mTORC1, STAT3, NF-κB and Notch signaling pathways, but also targets mitochondria in cancer cells to induce cell cycle arrest, growth inhibition and apoptosis. A number of studies have established the anticancer activities of niclosamide in both in vitro and in vivo models. Moreover, the inhibitory effects of niclosamide on cancer stem cells provide further evidence for its consideration as a promising drug for cancer therapy. This article reviews various aspects of niclosamide as they relate to its efficacy against cancer and associated molecular mechanisms.
20) Hospitals struggling against ‘C. diff’ bacteria Philly.com
October 7, 2016
21)
PHIL ID #9999 Photo Credit: Janice Haney Carr, Centers for Disease Control and Prevention. Download High Resolution. Description: This micrograph depicts gram-positive Clostridium difficile bacteria from a stool sample culture obtained using a .1µm filter.
The post Clostridia Difficile Enterocolitis Causes Prevention and Treatment appeared first on Jeffrey Dach MD .









Leave a Comment
You must be logged in to post a comment.