The Failure of Global Polio Eradication
My wife loaned me an old copy of Jane Goodall’s 1971 book,” In the Shadow of Man, about wild chimpanzees of the Gombe National Park. It was fascinating reading. However, I was saddened by an apparent polio outbreak in the chimpanzees which took place around 1966.(1)
One day, a beloved chimpanzee appeared in camp dragging a lifeless limb. Jane assumed it was polio, since an outbreak had been reported in a nearby village. They looked on hopelessly as one after another of the chimps were stricken with paralysis, eventually affecting about half dozen. They then tried giving the chimps oral polio vaccine as a preventive measure. In spite of their efforts, two of the chimps had to be euthanized with the camp pistol.
Unanswered Questions
There are many unanswered questions. For example, we don’t know if the causative agent was a poliovirus, coxsackie or some other entero-virus. If a polio virus was present, we assume it came from humans, yet we don’t know if it was wild type polio, or vaccine derived polio (VAPP.)(1) Perhaps the acute flaccid paralysis was caused by exposure to neurotoxic pesticide at the camp?
In Zaire in 1964, there was a similar outbreak of flaccid paralysis in chimpanzees. Observers were not sure if the etiology was a neuro-virus such as polio transmitted by humans, or exposure to neuro-toxic agricultural chemical sprays.(18) (limb paresis polio chimpanzees 1964 Kortlandt Pan African News )
Polio Vaccine Experimentation in Africa
In the preceding decade in the 1950’s, there had been local vaccine experimentation with live polio virus by Hilary Koprowski in both primates and villagers in the Belgian Congo.(20-23) So, surely, there was plenty of opportunity for a virulent vaccine derived polio virus to escape and circulate in the human population.(44) Although Koprowski’s publications contained glowing reports, his vaccine was never approved for human use.
Polio Vaccine Derived Paralytic Polio in Nigeria
In the 2011 Journal of Infectious Disease, Dr Wassilak from the CDC reported 336 cases of paralytic polio in Nigeria caused by the oral polio vaccine itself, which had mutated back to a virulent form. Similar cases of vaccine derived virus paralytic polio have been reported in the Belgian Congo (41-44) (see: Outbreak of vaccine-derived poliovirus in Nigeria.)
Vaccine Derived Polio in an Amish Community
This Jane Goodall chimpanzee polio outbreak reminded me of a 2005 report of an outbreak of Vaccine Derived Polio in an unvaccinated Amish community in Minnesota.(34-35)(13) Thankfully, in this human population, there was no paralysis, merely documentation of virus shedding in the stools. Molecular biology tools using reverse-transcriptase polymerase chain reaction (RT-PCR) assays, showed the virus was derived from an oral polio vaccine which had circulated in an unvaccinated community.(34-35)(13)
The Oral Polio Vaccine by Albert Sabin
 I was a kid in third grade in 1958 when the whole school was lined up in the hallway. Each one of us was given a sugar cube impregnated with oral polio vaccine. This was the Polio Vaccine developed by Albert Sabin, a live attenuated virus which causes infection in the gut, but is not neuro-virulent. The recipient develops immunity to the live attenuated virus, which is also protective for the wild type polio virus. Left image oral polio vaccine applied to sugar cubes courtesy of wikimedia.
I was a kid in third grade in 1958 when the whole school was lined up in the hallway. Each one of us was given a sugar cube impregnated with oral polio vaccine. This was the Polio Vaccine developed by Albert Sabin, a live attenuated virus which causes infection in the gut, but is not neuro-virulent. The recipient develops immunity to the live attenuated virus, which is also protective for the wild type polio virus. Left image oral polio vaccine applied to sugar cubes courtesy of wikimedia.
Houston, We Have a Problem
It was soon recognized that this harmless oral polio vaccine would occasionally revert back into a neurovirulent strain, causing paralytic polio in the recipient, as well as contacts exposed to shedding virus. Here is a quote from Dr. Hull:“Mutated Vaccine Derived Polio Virus that are neurovirulent in animal models can be isolated from the stools of children within days of vaccination” (10)
In 1993, it was discovered that attenuated polio vaccine reverts to its neuro-virulent form with mutations at only two positions. (12)
“Site-directed mutagenesis of P2/Sabin showed that mutations at just two positions, at 481 in the 5′ non-coding region and at VP1-143 in the capsid proteins, resulted in a highly neurovirulent virus”(12)
This is the reason why the CDC discontinued use of the live oral vaccine fifteen year ago in the US (in 2000), and switched to the killed inject-able Salk vaccine. Left image Scanning Electron Microscope of polio virus courtesy of polio.org
Global Polio Eradication With the Live Oral Vaccine
 The experience in India with the live oral polio vaccine is illustrative of the problems encountered with global polio eradication. (5)(36)
The experience in India with the live oral polio vaccine is illustrative of the problems encountered with global polio eradication. (5)(36)
Although India was declared “polio-free” with the last case reported January 13, 2011, the numbers of cases of acute flaccid paralysis in children has skyrocketed throughout the campaign. Left Image courtesy of Health India.
Here is a quote from Dr. Kaur 2014.(31)
The National Polio Surveillance Project data show that the polio eradication program has increased paralysis among children—from 3,047 cases yearly in 1997 to 61,038 cases in 2012, most now being classified as AFP instead of polio.(31) (note AFP=Acute Flaccid Paralysis)
Here we have “successfully” eradicated wild type polio from a country,(India) with the use of oral polio vaccine, yet the number of cases of acute flaccid paralysis has increased 20-fold. This is not success. This is failure. This type of self delusion is typical of government agencies run by morons, however to the most casual observer, this is an obvious farce.
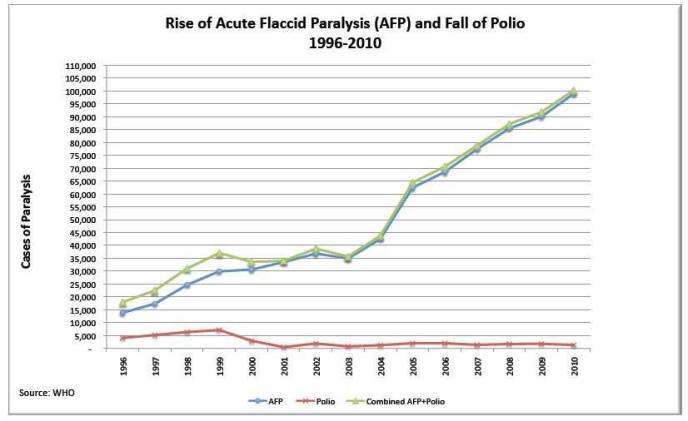
Global Eradication Failure in One Chart – below chart courtesy of the World Health Organization showing rise of Acute Flaccid Paralysis (blue line) while detectable polio virus cases (red line) decline. The live oral vaccine works quite well at eradicating detectable wild type polio virus, however the live oral vaccine is a failure at reducing numbers of children with flaccid paralysis.
Again, there are many unanswered questions. What is causing these increasing numbers of acute flaccid paralysis in India? Could it be possible that the polio virus is the wrong target, and that acute flaccid paralysis is being caused by some other infectious or non-infectious agent, ? Polio virus is only one of the many enteric viruses which can cause flaccid paralysis. Are there other entero-viruses involved, and which ones?
Neurotoxic Chemicals in Environment?
Are there environmental causes of flaccid paralysis that have been overlooked? Neurotoxic chemicals may cause acute flaccid paralysis. For example, exposure to DDT or other organo-phosphate insecticides can cause neurological disease resembling polio. (2,3) DDT was banned in the US in 1972, yet is still widely used in India. What is the exposure history for these effected children? And what about measuring blood and urine levels of DDT and other toxic chemicals in affected children? None of these studies have been done, so we don’t know. Above left image: Spraying DDT Courtesy of Universal News.
Vitamin C as an Anti-Toxin and Antiviral Agent
It is well known that children with nutritional deficiencies are more susceptible to infection as well as damaging effects of environmental toxins. In view of past research showing success with vitamin C as anti-toxic and anti-viral, wouldn’t it be prudent to conduct studies of vitamin C levels on the children with acute flaccid paralysis?(3) Do they have low vitamin C levels? Would vitamin C, in the form of fresh fruits and vegetables, or even supplements, reduce the incidence of acute flaccid paralysis? These are unanswered questions which await further study.
Failure Is Only the Beginning to Find the Real Answers
Global polio eradication campaigns with oral polio vaccine have eradicated wild type polio, yet have been a failure in terms of increasing acute flaccid paralysis cases in children. Case numbers have increased dramatically in spite of oral polio vaccination programs. This is what i call, “The Failure of Global Polio Eradication.” (37) (Polio eradication: a failure by Paul Yash)
A Wake Up Call
This failure is a Wake Up call to re-examine what we are doing, and ask some fundamental questions about the etiology of acute flaccid paralysis in children. Spending six billion dollars on a live polio vaccine against the wild type polio virus is obviously not the answer.(36) Let us spend the money on the right questions, so we can do the right studies and find the right answers.
Articles with related interest:
Spraying DDT to Eradicate Polio
Dissolving Illusions Book Review
Great Women in Medicine Elizabeth Kenny
Jeffrey Dach MD
7450 Griffin Road
Suite 180/190
Davie, Florida 33314
954-792-4663
Links and References:
For a list of articles on Vaccine Derived Polio Virus: Click Here
1) Jane Goodall In the Shadow of Man
Goodall, Jane. In the shadow of man. Houghton Mifflin Harcourt, 2000.
2) http://www.vaccinationcouncil.org/2011/11/17/smoke-mirrors-and-the-disappearance-of-polio/
Smoke, Mirrors, and the “Disappearance” Of Polio– November 17, 2011
Suzanne Humphries, MD
3) http://orthomolecular.org/resources/omns/v09n16.shtml
Orthomolecular Medicine News Service, August 7, 2013
Vitamin C and Polio. The Forgotten Research of Claus W. Jungeblut, M.D.
by Andrew W. Saul Editor, Orthomolecular Medicine News Service
With more time, I would find that his live oral vaccine had become the leading cause of polio in the US. What surprised me most was that the strongest criticism originated from the most eminent of sources: the other polio hero, Dr. Jonas Salk. On September 24, 1976, the Washington Post reported Dr. Salk’s assertion that the Sabin live oral virus vaccine had been the “principal if not sole cause” of every reported polio case in the United States since 1961. (1) Salk repeated this accusation July 6, 1977, when he was interviewed on CBC television (2), saying: “(W)e have known now since 1961 in the United States, and prior to that in other countries, that the live virus vaccine for polio does cause the disease itself.”
In 1996, one year after Salk died, the US Centers for Disease Control began a turn-away from the oral live vaccine and recommended killed virus injections for the first two rounds of infant polio immunization. By 2000, CDC stated that “To eliminate the risk for vaccine-associated paralytic poliomyelitis, an all-Injected Polio Virus schedule is recommended for routine childhood vaccination in the United States.” (3) Thus only after two decades would orthodoxy at last take heed of the cautionary words of Dr. Salk, the man credited with creating the first polio vaccination.
——————-
4) http://www.dailysabah.com/health/2015/10/24/shift-in-vaccine-use-needed-for-polio-free-world-by-2019-who-experts
Shift in vaccine use needed for polio-free world by 2019: WHO experts
FRENCH PRESS AGENCY – AFP
2015 India
5) http://www.rajasthannews1.com/2015/10/india-is-polio-free-nation-but-still.html
India Is Polio Free Nation: But, Still Suffers from 18,000 cases of Non Polio Acute Flaccid Paralysis Published By: Rajasthan News1 on: Friday, October 23, 2015
It’s been three years since the last case was reported. The World Health Organization declared India ‘Polio-Free’ in 2014 which is a significant achievement in the history of Public Health.
Dr. Gupta expressed that, “In spite of the World Health Organization declaring India a Polio- Free of the ‘Wild Type Polio’ virus, the country has seen an increase in the cases of Non-Polio Paralysis which is a huge cause of concern. According to a report, in 2004, 12,000 cases of non-polio paralysis were reported which increased by 2012 to 53,563 cases for a national rate of 12 per 100,000 children.”
2003 the OPV Paradox
6) http://www.ncbi.nlm.nih.gov/pubmed/12931339
Rev Med Virol. 2003 Sep-Oct;13(5):277-91.
Polio eradication: the OPV paradox.
Dowdle WR1, De Gourville E, Kew OM, Pallansch MA, Wood DJ.
Routine and mass administration of oral polio vaccine (OPV) since 1961 has prevented many millions of cases of paralytic poliomyelitis. The public health value of this inexpensive and easily administered product has been extraordinary. Progress of the Global Polio Eradication Initiative has further defined the value of OPV as well as its risk through vaccine-associated paralytic poliomyelitis (VAPP) and vaccine-derived polioviruses (VDPV). Although both are rare, once wild poliovirus transmission has been interrupted by OPV, the only poliomyelitis due to poliovirus will be caused by OPV. Poliovirus will be eradicated only when OPV use is discontinued. This paradox provides a major incentive for eventually stopping polio immunization or replacing OPV, but it also introduces complexity into the process of identifying safe and scientifically sound strategies for doing so. The core post eradication immunization issues include the risk/benefits of continued OPV use, the extent of OPV replacement with IPV, possible strategies for discontinuing OPV, and the potential for development and licensure of a safe and effective replacement for OPV. Formulation of an informed post eradication immunization policy requires careful evaluation of polio epidemiology, surveillance capability, vaccine availability, laboratory containment, and the risks posed by the very tool responsible for successful interruption of wild poliovirus transmission.
2005 Vsaccine Derived Polio-virus
7) http://www.ncbi.nlm.nih.gov/pubmed/16153180
Annu Rev Microbiol. 2005;59:587-635.
Vaccine-derived polioviruses and the endgame strategy for global polio eradication. Kew OM1, Sutter RW, de Gourville EM, Dowdle WR, Pallansch MA.
As the global eradication of wild poliovirus nears, the World Health Organization (WHO) is addressing challenges unprecedented in public health. The live, attenuated oral poliovirus vaccine (OPV), used for more than four decades to interrupt poliovirus transmission, and the vaccine of choice for developing countries, is genetically unstable. Reversion of the small number of substitutions conferring the attenuated phenotype frequently occurs during OPV replication in humans and is the underlying cause of the rare cases of vaccine-associated paralytic poliomyelitis (VAPP) in OPV recipients and their close contacts. Whereas VAPP has long been recognized, two other adverse events have been identified more recently:
(a) long-term excretion of highly evolved vaccine-derived polioviruses (VDPVs) in persons with primary immunodeficiencies, and
(b) polio outbreaks associated with circulating VDPVs in areas with low rates of OPV coverage. Developing a posteradication strategy to minimize the risks of VDPV emergence and spread has become an urgent WHO priority.
2007 polio mutates from Coxsakhie virus – impossible to eradicate this
8) http://www.pnas.org/content/104/22/9457.full
Evidence for emergence of diverse polioviruses from C-cluster coxsackie A viruses and implications for global poliovirus eradication Ping Jiang *, Johan A. J. Faase † , Hidemi Toyoda *, Aniko Paul *, Eckard Wimmer * , ‡ , and Alexander E. Gorbalenya † Edited by Robert G. Webster, St. Jude Children’s Research Hospital, Memphis, TN, and approved April 12, 2007 (received for review January 18, 2007)
The poliovirus (PV) eradication campaign is conducted on the premise that this virus, because of the lack of a zoonotic reservoir, will not reemerge once eradicated. This report examines the origin of PV using theoretical and experimental approaches. Our rooted phylogenetic analysis suggests a speciation of PV from a C-cluster coxsackie A virus (C-CAV) ancestor through mutation of the capsid that caused a receptor switch from intercellular adhesion molecule-1 to CD155, leading to a change of pathogenicity. This hypothesis is supported experimentally with chimeras generated from three different pairs of PV and C-CAV. Those carrying the PV capsid and the replication proteins of C-CAVs replicated well, whereas their reciprocal counterparts were either debilitated or dead. This phenomenon of asymmetry is observed also in recombinants between PV1 and C-CAV20, selected in tissue culture cells using a previously undescribed protocol. The recombinants are generated at frequencies of 10−6 typical for PV interserotype recombination. Strikingly, they resemble genetically and phenotypically, including neurovirulence in CD155 transgenic mice, the large majority of circulating vaccine-derived PVs that have caused poliomyelitis outbreaks in different parts of the world. These data provide experimental evidence for C-CAVs being partners to PVs in generating diverse PV progeny by homologous recombination. They support speciation of a novel human pathogen (PV) from a pool of different human pathogens (C-CAVs).
In a PV-free world without PV neutralizing antibodies, contemporary C-CAV, like their ancestor(s), could be fertile ground for a PV-like agent to emerge by mutation.
2011
—————————–
These circulating VDPVs (cVDPVs) have recently caused iatrogenic epidemics of paralytic poliomyelitis in 15 regions of the world, including Madagascar in 2002 and 2005
9) http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3185806/
Combelas, Nicolas, et al. “Recombination between Poliovirus and Coxsackie A Viruses of Species C: A Model of Viral Genetic Plasticity and Emergence.” Viruses 3.8 (2011): 1460.
free full pdf
These circulating VDPVs (cVDPVs) have recently caused iatrogenic epidemics of paralytic poliomyelitis in 15 regions of the world, including Madagascar in 2002 and 2005 (see below Section 4 and Figure 2). Most cVDPVs are recombinants between PV and other HEV-C, principally coxsackie A viruses (CAV). All cVDPVs have similar genomic features: the region encoding the capsid proteins is from vaccine strains, but with more than 1% nt substitutions, and some or all of the rest of the genome, particularly the region encoding nonstructural proteins, originates from other HEV-C. Thus, cVDPVs greatly complicate the implementation of surveillance and vaccination strategies aimed at eradicating poliomyelitis and PV.
The global public health program consisting of vaccination of the human population with OPV has been quite successful. However, the emergence of cVDPVs is one of the major issues threatening the benefit of the eradication program. In fact, the spreading of a huge amount of man-made attenuated vaccine virus in the human population allowed us to discover an unknown interaction between PV and the members of its natural ecological niche. This discovery is of tremendous importance in terms of medical as well as basic virology and underlines the interest to keep close contact between basic and medical sciences, in particular when viral human pathogens are concerned.
————————–
1999
pdf free
10) Hull, Harry F., and R. Bruce Aylward. “Invited commentary: The scientific basis for stopping polio immunization.” AMERICAN JOURNAL OF EPIDEMIOLOGY. 150.10 (1999): 1022-5.
Mutated VDPV that are neurovirulent in animal models can be isolated from the stools of children within days of vaccination (14).
The meeting concluded that all vaccination against polio can and should stop.
Pending the results of current research, simply stopping the use of oral poho vaccine continues to be the most likely strategy.
2004 free pdf
11) Minor, Philip D. “POLIO ERADICATION, CESSATION OF VACCINATION AND RE-EMERGENCE OF DISEASE.” NATURE REVIEWS| MICROBIOLOGY 2 (2004): 473.
On rare occasions vaccine strains cause poliomyelitis in recipients or
their contacts, owing to reversion of mutations that attenuate virulence during growth in the vaccinee.
All cVDPV strains that have been characterized so
far are composed of the structural proteins of one of the
vaccine strains, whereas the rest of the virus derives
from so-far unidentified enteroviruses from the same
virus group as poliovirus12 (BOX 1). It is thought
that recombination between the vaccine strain and
an enterovirus is responsible for emergence of the
cVDPV strains that have been identified so far. For
each cVDPV outbreak that has been analysed the
recombinant enterovirus partner is different 9–11, and in
Haiti, the Dominican Republic and Madagascar there
was evidence for multiple recombination events with
different enterovirus partners 9,11.
The occurrence of VAPP, iVDPVs and cVDPVs
indicates that vaccine strains can regain virulence,
transmit between individuals with low or high efficiency,
or persist in an individual without being recognized
for weeks to years. Once the wild-type virus is
eradicated, the only cause of paralytic polio disease will
be vaccine-derived strains.
Eradication of poliomyelitis disease will only be
successful if the poliovirus is eradicated. The main
strategy that has been used so far is the live attenuated
poliovirus vaccine developed by Sabin, which is known
to revert to produce a virulent and transmissible virus
— albeit at low frequency.
1993
12) http://www.ncbi.nlm.nih.gov/pubmed/8390752
Virology. 1993 Jan;192(1):18-26.
Genetic basis of attenuation of the Sabin type 2 vaccine strain of poliovirus in primates. Macadam AJ1, Pollard SR, Ferguson G, Skuce R, Wood D, Almond JW, Minor PD.
The type 2 live-attenuated vaccine strain of poliovirus (P2/Sabin) is associated with rare cases of poliomyelitis in vaccinees or their contacts. Recombinants were generated between infectious clones of a neurovirulent isolate from one such case (P2/117) and P2/Sabin and neurovirulence assays suggested that a maximum of six nucleotide differences between the two strains were responsible for their phenotypic difference. Site-directed mutagenesis of P2/Sabin showed that mutations at just two positions, at 481 in the 5′ non-coding region and at VP1-143 in the capsid proteins, resulted in a highly neurovirulent virus. Other nucleotide changes may have weaker phenotypic effects. These results are consistent with those reported in the mouse model by Ren et al. [J. Virol. 65, 1377, (1991)] indicating that, for P2/Sabin at least, the same determinants of attenuation are important in both primates and transgenic mice expressing the poliovirus receptor. Sequence analysis of isolates from other vaccine-associated cases of poliomyelitis and from healthy vaccinees showed that both major determinants of attenuation are unstable on human passage, although selection pressures against an A at 481 are stronger than those against an Ile at 1143.
mutations at just two positions, at 481 in the 5′ non-coding region and at VP1-143 in the capsid proteins, resulted in a highly neurovirulent virus.
2005 – Polio in unvaccinated AMISH – CDC
13) http://www.cdc.gov/mmwr/preview/mmwrhtml/mm54d1014a1.htm
Poliovirus Infections in Four Unvaccinated Children — Minnesota, August–October 2005. On September 29, 2005, the Minnesota Department of Health (MDH) identified poliovirus type 1 in an unvaccinated, immunocompromised infant girl aged 7 months (the index patient) in an Amish community whose members predominantly were unvaccinated for polio. The patient has no paralysis; the source of the patient’s infection is unknown. Subsequently, poliovirus infections in three other children within the index patient’s community have been documented. This report summarizes the ongoing investigation, provides information regarding poliovirus exposure risks and prevention measures in the United States, and offers recommendations to state health departments and clinicians.
2009
full free
14) http://cid.oxfordjournals.org/content/49/8/1287.full
Shahzad, Aamir. “Time for a worldwide shift from oral polio vaccine to inactivated polio vaccine.” Clinical infectious diseases 49.8 (2009): 1287-1288.
A major concern about OPV is its ability to revert to a form that can cause paralysis. Outbreaks of vaccine-associated paralytic poliomyelitis (VAPP) have been reported in many countries of the world [2, 4].
In 2005, it was reported that children in a small village in the United States had contracted vaccine-derived polio. In Nigeria, >70 cases have been reported. In 2006, ~1600 cases of vaccine-induced polio occurred in India, according to the Indian Medical Association Sub-Committee on Immunisation’s report on the Polio Eradication Initiative [3]. The point to be noted is that these cases were reported during repeated mass-immunization campaigns in which repeated doses of OPV were administered.
The above observations suggest that OPV has lost its effectiveness in providing herd immunity. It seems that children are getting polio from OPV, and it also seems that OPV is proving to be ineffective in stopping polio transmission from another source. Therefore, the whole world—and especially developing countries—should shift from OPV to IPV, in my opinion.
2010
15) http://www.nejm.org/doi/full/10.1056/NEJMp1005405
The Bumpy Road to Polio Eradication John F. Modlin, M.D. N Engl J Med 2010;
The emergence of cVDPVs forces us to accept the reality that we are fighting fire with fire and that once eradication of WPV is assured, the use of live poliovirus vaccines will need to cease globally in a coordinated manner. Because cVDPVs will probably continue to circulate for at least 1 to 3 years after WPVs are eradicated,4
2010
full pdf
16) Jenkins, Helen E., et al. “Implications of a circulating vaccine-derived poliovirus in Nigeria.” New England Journal of Medicine 362.25 (2010): 2360-2369.
!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
2013
17) http://rstb.royalsocietypublishing.org/content/368/1623/20120140
Grassly, Nicholas C. “The final stages of the global eradication of poliomyelitis.” Philosophical Transactions of the Royal Society of London B: Biological Sciences 368.1623 (2013): 20120140.
Approximately one case of vaccine-associated paralytic poliomyelitis (VAPP) occurs per 750 000 doses of trivalent OPV for the first dose given [10], compared with one case of poliomyelitis per 100 to 1000 infections with wild-type poliovirus, depending on the serotype [11–13].
(a) Vaccine-derived polioviruses and OPV cessation
The Sabin poliovirus strains in use today were attenuated through an empirical process involving serial passage in cell culture and primates and assessment of neuropathogenicity in monkeys [4]. The molecular basis of attenuation was only elucidated more recently. A series of studies revealed just a small number of mutations were responsible for the reduced neuropathogenicity of each of the Sabin strains (reviewed in [80]). Furthermore, following administration of OPV, Sabin poliovirus rapidly evolves through a process of mutation and intertypic recombination to lose these attenuating mutations [81,82]. Loss of attenuating mutations is frequently found among vaccine polioviruses isolated from children with VAPP, following administration of OPV [83,84]. The problem of VAPP was recognized within a year of licensure of OPV [85]. It also became apparent that VAPP could occur among contacts of individuals who had received OPV as a result of secondary spread of the vaccine poliovirus. Indeed, the number of children with suspected ‘contact’ VAPP is typically equivalent to or can exceed the number of cases of ‘recipient’ VAPP [86,87].
The theoretical possibility of outbreaks of poliomyelitis as a result of sustained transmission of vaccine-derived polioviruses has long been recognized [2]. This risk remained theoretical until the year 2000, when genetic sequence analysis of poliovirus isolated during an outbreak of poliomyelitis in Haiti and the Dominican Republic found a serotype 1 Sabin origin for the virus [7]. Recognition of this outbreak of vaccine-derived poliovirus (termed VDPV) was followed by further outbreaks of independently evolved VDPVs in other countries and by retrospective identification of VDPVs from stored stool samples associated with past outbreaks of poliomyelitis [88,89]. Improved laboratory methods based on real-time PCR with primers specific to nucleotide substitutions typically observed early in VDPV evolution are now used widely within the GPLN to identify emergent and circulating VDPV from stool and environmental samples [90]. The GPEI labels a vaccine-derived poliovirus as ‘circulating’ (a cVDPV) when it is at least 1 per cent divergent from Sabin in the VP1 region (0.6% for serotype 2) and where at least two children with AFP shed closely related virus. At the time of writing 20 cVDPV had been reported from 20 different countries, the vast majority derived from the serotype 2 Sabin strain [91] and many recombinants with other human enteroviruses [92,93]. The predominance of serotype 2 VDPV is the result of the greater fitness of this virus compared with the other Sabin strains and declines in population immunity to serotype 2 following the introduction of monovalent and bivalent OPVs.
In a study of the largest outbreak recorded to date, serotype 2 VDPV in Nigeria was found to have a similar attack rate to co-circulating wild-type polioviruses [8]. Similar results were found for an outbreak of a serotype 1 VDPV in Indonesia [94]. It is therefore clear that VDPVs can evolve to regain equivalent transmissibility and pathogenicity to wild-type polioviruses, and outbreaks must be responded to with the same vigour.
The identification of poliomyelitis outbreaks caused by VDPVs made apparent the risks of continued OPV use post-eradication of wild-type polioviruses. At a consultation held by the WHO in 2003, it was agreed that OPV use would have to cease post-eradication and that a clear ‘endgame’ strategy was needed [95]. Coordinated global cessation of OPV, 3 years after the last wild-virus-associated case of poliomyelitis was identified as a strategy that could minimize risks by stopping the introduction of vaccine poliovirus into the environment at a time when population immunity and the sensitivity of AFP surveillance are at their maximum [96]. Experience from Cuba and Mexico had shown that after mass immunization with trivalent OPV, and in the absence of routine vaccination with OPV, Sabin polioviruses disappeared from stool and environmental samples within 4–5 months [97–99]. More recently, countries switching to routine immunization with IPV have also documented disappearance of Sabin polioviruses several months after national immunization days or cessation of OPV [100,101].
In 2011, a revised endgame strategy was proposed by the GPEI, allowing for phased removal of Sabin poliovirus serotypes [102]. Over the last decade just over 500 children had been reported with poliomyelitis as a result of serotype 2 cVDPVs (approximately 90% of all documented poliomyelitis cases due to cVDPVs) and more than 1500 children were estimated to have developed VAPP as a result of the serotype 2 component of the trivalent vaccine [91,103]. Yet circulation of serotype 2 wild-type poliovirus was last recorded in 1999. Removal of serotype 2 Sabin poliovirus from all routine and SIAs would prevent these cases of poliomyelitis while providing an opportunity to eradicate one of the three poliovirus serotypes at a time of heightened surveillance and outbreak response capacity. This accelerated endgame strategy could begin as soon as currently circulating serotype 2 VDPVs are eliminated, raising the possibility of global eradication of at least one poliovirus serotype in the near future.
1964 chimpanzees
18) Epidemic limb paresis poliochimpanzees 1964 Kortlandt Pan African News Kortlandt, Adriaan. “Letters: An epidemic of limb paresis (polio?) among the chimpanzee population at Beni (Zaire) in 1964, possibly transmitted by humans (Additional information to Pan Africa News 2 (2), 1995).” (1996).
2010 chimps
full pdf
19) poliovirus antibody orally vaccinated chimpanzees Uganda Mugisha 2010
Mugisha, Lawrence, et al. “Evaluation of poliovirus antibody titers in orally vaccinated semi-captive chimpanzees in Uganda.” Journal of medical primatology 39.2 (2010): 123-128.
Polio Vaccine koprowski africa
20) http://vaxtruth.org/2012/04/the-polio-vaccine-part-3-2/
Koprowski’s vaccine was not approved for human use, so it was discontinued in 1960 following the African trials [100]. Thus, it was only administered to inhabitants of the Belgian Congo, Rwanda and Burundi [104,121
1958 Belgian COngo Live polio virus vaccine
21) Courtois, Gh, et al. “Preliminary report on mass vaccination of man with live attenuated poliomyelitis virus in the Belgian Congo and Ruanda-Urundi.” British medical journal 2.5090 (1958): 187.
1960 Congo
22) Lebrun, André, et al. “Vaccination with the CHAT strain of type 1 attenuated poliomyelitis virus in Léopoldville, Belgian Congo: 1. Description of the city, its history of poliomyelitis, and the plan of the vaccination campaign.” Bulletin of the World Health Organization 22.3-4 (1960): 203.
1960 Koprowski full pdf
23) Koprowski H. Historical aspects of the development of live virus vaccine in poliomyelitis. British Medical Journal, 1960;ii:85–91.
Koprowski, Hilary. “Historical aspects of the development of live virus vaccine in poliomyelitis.” British Medical Journal 2.5192 (1960): 85.
——————-
humans
24) http://www.biomedcentral.com/1471-2334/2/2
Immunity status against poliomyelitis in Germany: Determination of cut-off values in International Units. Sabine Diedrich*, Hermann Claus and Eckart Schreier Robert Koch Institute, Nordufer 20, 13353 Berlin, Germany
BMC Infectious Diseases 2002, 2:2
The last indigenous case of poliomyelitis was diagnosed in Germany in 1990. The last imported wild viruses were detected in patients with travel history to India and Egypt in 1992.
Since that period of time there were found only 0 – 3 cases of vaccine-associated paralytic poliomyelitis (VAPP) per year. In order to prevent these VAPP – cases the vaccination strategy has been changed in Germany. Since 1998 it is recommended to use only the inactivated vaccine (IPV).
The goal of eradication of poliomyelitis has been complicated by some events in 2000–2001. First, the discovery of circulation of a vaccine virus strain that mutated to regain its disease-causing ability in Hispaniola. This outbreak in the Dominican Republic and Haiti was the first in the Americas since 1991 and occurred in areas of very low vaccination coverage [7]. Second, the polio cases in Bulgaria. There had been no reported cases of polio in Europa since November 1998. In spring 2001 two gypsy unvaccinated children were paralyzed by a wild poliovirus in Bulgaria, which was imported from India [1].
25) http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3782271/
Baicus, Anda. “History of Polio Vaccination.” World Journal of Virology 1.4 (2012): 108–114. PMC. Web. 2 Oct. 2015.
26) http://www.nejm.org/doi/full/10.1056/NEJMp1005405
The Bumpy Road to Polio Eradication John F. Modlin, M.D. N Engl J Med 2010; 362:2346-2349June 24, 2010
27) http://articles.latimes.com/2011/feb/09/opinion/la-oe-orent-polio-20110209
Op-Ed The polio virus fights back Sabin’s oral vaccine is actually causing new outbreaks of the disease. by February 09, 2011|By Wendy Orent
!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
28) Diamond, Bruce. “Global polio campaign doomed to fail, experts warn.” Nature medicine 11.12 (2005): 1260-1260.
Virus shed by vaccinated individuals could keep the virus in circulation for decades. Five children in a secluded Amish village in the US contracted vaccine-derived polio, scientists announced in October, baffling epidemiologists about the infection’s origin. But some experts say the episode is a clear warning that the global polio eradication campaign is unrealistic.
—————–
2006
India 2006
full pdf
29) Polio Eradication Experts Misled Us Medical Veritas 2006 Paul Yash
by Paul Yash in Bani Park. “Polio eradication: experts have misled us.” Medical Veritas: The Journal of Medical Truth 3.1 (2006): 781.
The most baffling aspects of this polio eradication program are:
(i) no one knows if this vaccine is providing protection,
(ii) there is unacceptably high incidence of VAPP and doctors are advised not to let people know that OPV can and is causing polio in many children, and
(iii) IPV is not being made avail-able in India even for those children who are immunocompro-mised due to disease or drugs or who have immunocompro-mised close contacts, thus putting many individuals at known higher risk to develop VAPP.
Pakistan 2015
30) http://jech.bmj.com/content/early/2015/07/31/jech-2015-205530.extract
Ahmad, Syed Osama, et al. “Pakistan: the final frontier for global polio eradication.” Journal of epidemiology and community health (2015): jech-2015.
workers killed
parents refuSe- arrested by military-
OPV causes vaccine associated paralytic polio (VAPP)
India 2014
!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
full pdf
31) Polio Eradication in India Kaer 2014
Kaur, Sharanjit, Anand Sharma, and Harinder Jot Singh. “Polio Eradication in India: Need to Switch over to IPV from OPV.” May 2014
Polio cases due to vaccine-associated paralytic
poliomyelitis (VAPP) and outbreaks due to
circulating vaccine-derived polioviruses (cVDPVs),
are the two main reasons for eventually stopping the
use of OPV for routine immunization in all countries.
The National Polio Surveillance Project data show
that the polio eradication program has increased
paralysis among children—from 3,047 cases yearly
in 1997 to 61,038 cases in 2012, most now being
classified as AFP instead of polio.19
Oral polio vaccines (OPV) contain live, weakened
virus, which can – on very rare occasions – revert to
a paralytic form and regain the ability to circulate.
Replacing OPV with inactivated polio vaccines
(IPV), which carry no risk of paralysis, is necessary
to ensure a polio free world.
———————
2014 latin america
32) http://jid.oxfordjournals.org/content/early/2014/02/10/infdis.jit602.full
Landaverde, J. Mauricio, et al. “Vaccine-Associated Paralytic Poliomyelitis in the Postelimination Era in Latin America and the Caribbean, 1992–2011.” Journal of Infectious Diseases (2014): jit602.
———–
monkey failure
33) Grossman, L. H., and S. D. Kramer. “Attempts to Infect the Common Marmoset Monkey with the Virus of Poliomyelitis.” Experimental Biology and Medicine 35.2 (1936): 345-347. From the Laboratories of the Infantile Paralysis Commission of the Long Island College of Medicine.Five common Marmoset monkeys were inoculated intracerebrally, intraperitoneally, and intranasally with 10 and 20% suspensions of active poliomyelitic cord. The monkeys remained unaffected by such inoculations. The common Marmoset does not therefore appear to be susceptible to infection with the virus of poliomyelitis.
34) Alexander, James P., et al. “Transmission of imported vaccine-derived poliovirus in an undervaccinated community in Minnesota.” Journal of Infectious Diseases 199.3 (2009): 391-397.
35) Polio Outbreak Occurs Among Amish Families In Minnesota
By David Brown Washington Post Staff Writer Friday, October 14, 2005
36) Vashisht, Neetu, and Jacob Puliyel. “Polio programme: let us declare victory and move on.” Indian J Med Ethics 9.2 (2012): 114-117.
37) Polio eradication failure Paul Yash 2006 Paul, Yash. “Polio eradication programme: A failure.” Economic and Political Weekly (2006): 4538-4540.
38) A Critique Of Scientific Literature – Pesticides And Polio by Jim West
39) Health-Wall Street Journal Jun 26, 2015
India’s Victory Over Polio Has an Unexpected Consequence By Atish Patel
40) The polio virus fights back. Sabin’s oral vaccine is actually causing new outbreaks of the disease. February 09, 2011|By Wendy Orent LA Times
41) Officials Say Drug Caused Nigeria Polio
By MARIA CHENG The Associated Press October 5, 2007
The “DRUG” is live oral polio vaccine.
42) Wassilak, Steven, et al. “Outbreak of type 2 vaccine-derived poliovirus in Nigeria: emergence and widespread circulation in an underimmunized population.” Journal of Infectious Diseases 203.7 (2011): 898-909.
An outbreak of infection involving 315 cases of type 2 circulating vaccine-derived poliovirus (cVDPV2; >1% divergent from Sabin 2) occurred during July 2005–June 2010, a period when 23 of 34 SIAs used monovalent or bivalent oral poliovirus vaccine (OPV) lacking Sabin 2. In addition, 21 “pre-VDPV2” (0.5%–1.0% divergent) cases occurred during this period. Both cVDPV and pre-VDPV cases were clinically indistinguishable from cases due to wild poliovirus.
43) Sabin vaccine reversion in the field: a comprehensive analysis of Sabin-like poliovirus isolates in Nigeria by Michael Famularea#, Stewart Changa, Jane Iberb, Kun Zhaob, Johnson A. Adenijic, David Bukbukd, Marycelin Babae, Matthew Behrendf, Cara C. Burnsb and M. Steven Obersteb
To assess the dynamics of genetic reversion of live poliovirus vaccine in humans, we studied molecular evolution in Sabin-like poliovirus isolates from Nigerian acute flaccid paralysis cases obtained from routine surveillance. We employed a novel modeling approach to infer substitution and recombination rates from whole-genome sequences and information about poliovirus infection dynamics and individual vaccination history. We confirmed observations from a recent vaccine trial that VP1 substitution rates are increased for Sabin-like isolates relative to the wild-type rate due to increased non-synonymous substitution rates. We also inferred substitution rates for attenuating nucleotides and confirmed that reversion can occur in days to weeks after vaccination. We combine our observations for Sabin-like evolution with the wild-type circulating VP1 molecular clock to infer that the mean time from the initiating vaccine dose to the earliest detection of circulating vaccine-derived poliovirus (cVDPV) is 300 days for type 1, 210 days for type 2, and 390 days for type 3. Phylogenetic relationships indicated transient local transmission of Sabin 3 and possibly Sabin 1 during periods of low wild polio incidence. Comparison of Sabin-like recombinants with known Nigerian VDPV recombinants shows that while recombination with non-Sabin enteroviruses is associated with cVDPV, the recombination rates are similar for Sabin-Sabin and Sabin-non-Sabin enterovirus recombination after accounting for time from dose to detection. Our study provides a comprehensive picture of the evolutionary dynamics of oral polio vaccine in the field.
44) Bull Soc Pathol Exot. 2015 Oct;108(4):235-241. Epub 2015 Oct 10.
Circulating vaccine-derived poliovirus type 2 outbreak in Democratic Republic of Congo 2011-2012. Bazira L1, Coulibaly T2, Mayenga M2, Ncharre C2, Yogolelo R3, Mbule A2, Moudzeo H2, Lwamba P2, Mulumba AW4, Cabore J2.
According to the WHO records of 2013, the incidence of poliomyelitis was reduced by more than 99%, the number of endemic countries decreased from 125 in 1988 to 3 in 2013 and over 10 million cases were prevented from poliomyelitis thanks to the intensive use of Oral polio vaccine (OPV). However, the emergence of circulating vaccine-derived poliovirus strains (cVDPV), causing serious epidemics like the wild poliovirus, is a major challenge on the final straight towards the goal of eradication and OPV cessation. This paper describes the cVDPVoutbreak that occurred in the Democratic Republic of Congo (DRC) from November 2011 to April 2012. All children under 15 years of age with acute flaccid paralysis (AFP) and confirmed presence of cVDPV in the stool samples were included. Thirty (30) children, all from the administrative territories of Bukama and Malemba Nkulu in the Katanga Province (south-east DRC), were reported. The virus responsible was the cVDPV type 2 (0.7% -3.5% divergent from the reference Sabin 2 strain) in 29 children (97%) and the ambiguous vaccine-derived poliovirus strain (0.7% divergent) was confirmed in one case (3%), a boy seventeen months old and already vaccinated four times with OPV. Twentyfive children (83%) were protected by any of the routine EPI vaccines and 3 children (10%) had never received any dose of OPV. In reaction, DRC has conducted five local campaigns over a period of 10 months (from January to October 2012) and the epidemic was stopped after the second round performed in March 2012. As elsewhere in similar conditions, low immunization coverage, poor sanitation conditions and the stop of the use of OPV2 have favoured the emergence of the third cVDPV epidemic in DRC. The implementation of the Strategic Plan for Polio eradication and endgame strategic plan 2013-2018 will prevent the emergence of cVDPV and set up the conditions for a coordinated OPV phase out.
Jeffrey Dach MD
7450 Griffin Road Suite 190
Davie, Fl 33314
954-792-4663
www.jeffreydachmd.com
http://www.drdach.com
http://www.naturalmedicine101.com
http://www.bioidenticalhormones101.com
http://www.truemedmd.com
Disclaimer click here: http://www.drdach.com/wst_page20.html
The reader is advised to discuss the comments on these pages with his/her personal physicians and to only act upon the advice of his/her personal physician. Also note that concerning an answer which appears as an electronically posted question, I am NOT creating a physician — patient relationship. Although identities will remain confidential as much as possible, as I can not control the media, I can not take responsibility for any breaches of confidentiality that may occur.
Copyright (c) 2015 Jeffrey Dach MD All Rights Reserved. This article may be reproduced on the internet without permission, provided there is a link to this page and proper credit is given.
FAIR USE NOTICE: This site contains copyrighted material the use of which has not always been specifically authorized by the copyright owner. We are making such material available in our efforts to advance understanding of issues of significance. We believe this constitutes a ‘fair use’ of any such copyrighted material as provided for in section 107 of the US Copyright Law. In accordance with Title 17 U.S.C. Section 107, the material on this site is distributed without profit to those who have expressed a prior interest in receiving the included information for research and educational purposes.
Serving Areas of: Hollywood, Aventura, Miami, Fort Lauderdale, Pembroke Pines, Miramar, Davie, Coral Springs, Cooper City, Sunshine Ranches, Hallandale, Surfside, Miami Beach, Sunny Isles, Normandy Isles, Coral Gables, Hialeah, Golden Beach ,Kendall,sunrise, coral springs, parkland,pompano, boca raton, palm beach, weston, dania beach, tamarac, oakland park, boynton beach, delray,lake worth,wellington,plantation.
The post The Failure of Global Polio Eradication appeared first on Jeffrey Dach MD .










Leave a Comment
You must be logged in to post a comment.